Catalysis for Sustainable Syntheses
Prof. Jagadeesh Rajenahally
The world has transformed significantly over the past and current century with notable advancements as a result of scientific discoveries and their applications. However, the growing population and rapid urbanization lead to higher resource and energy consumption, which poses significant challenges in creating a sustainable society, particularly concerning health, energy, and the environment. In this regard, achieving sustainability and enabling a circular economy are major goals, which are crucial for ensuring sufficient availability of resources, mitigating environmental problems, and lowering energy demands. To achieve these goals, sustainable and circular chemistry play a dominant role by transition from a linear take-make-dispose model to one that prioritizes resource efficiency and conservation, use of renewable feedstocks, waste reduction, and the continuous reuse of materials. In particular, the use of abundant and renewable resources and the recycling/upscaling of waste materials are essential aspects as they reduce/avoid dependence on non-renewable (fossil-based) resources and enable a circular model with a closed-loop economy. Notably, the applicability of suitable chemical synthetic processes (organic synthetic methodologies) is fundamental prerequisite for achieving sustainable and circular chemistry by facilitating the development of eco-friendly products, employing renewable raw materials, and designing a closed-loop system though green synthesis. To perform chemical synthesis in more sustainable, waste-free, economically viable and environmentally friendly manner, applicability of catalytic processes is crucial. Thus, catalysis is essential for achieving sustainable and circular chemistry by enabling cost-effective and more efficient chemical synthesis involving the valorization of renewable feedstocks and conversion of waste-materials and use them as resources for chemical upgrading.
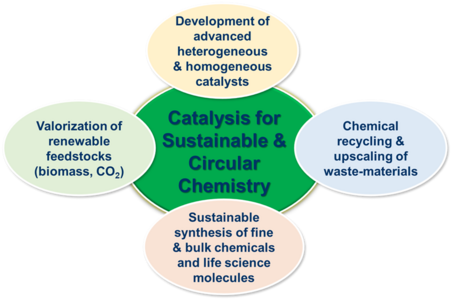
Given the importance of sustainable and circular chemistry, our group’s research focuses on the development of environmentally beneficial, economically viable and industrially feasible catalytic processes for advanced organic synthesis, valorization of renewable feedstocks and recycling/upscaling of waste materials.
Our research focuses on:
(1) Catalyst development (both heterogeneous and homogeneous catalysts).
(2) Catalysis for sustainable and advanced organic synthesis (Sustainable catalytic processes for essential fine and bulk chemicals, pharmaceuticals, agrochemicals and biomolecules).
(3) Catalytic valorization of renewable feedstocks (biomass, CO2 …) to produce value-added chemicals and fuels.
(4) Chemical recycling/upscaling of waste-materials (plastics- and bio-waste) and use them as resources for enabling circular economy.
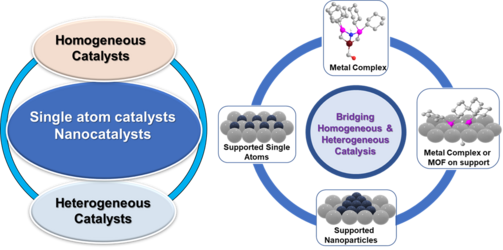
Catalysis is a key technology that governs the chemical processes with extensive applications in the production of all kinds of chemicals and life science molecules as well as daily life products. Thus, the modern chemical synthesis and industrialized world mainly relies on catalysis and would be inconceivable without the applicability of catalytic processes. As a result, >80% of chemicals produced globally are manufactured using catalytic processes. Thus, the development of a ‘right’ and ‘ideal’ catalytic system is crucial. We focus on the development of both heterogeneous and homogeneous catalysts for organic synthesis, industrial processes, valorization of renewable feedstocks and conversion of waste-materials. Homogeneous complexes are highly selective and active and but not very stable and are difficult to recycle or reuse. On the other hand, traditional heterogeneous materials are quite stable and can be conveniently recyclable, but these ones exhibit less selectivities and activities. Considering the individual features of these catalysts, it is difficult to achieve an “ideal” catalytic system, which should be very stable, easily recyclable and reusable, as well as highly active and selective. Thus, the creation of synergies between homogeneous and heterogeneous catalysis aspects is highly essential to design an ‘ideal’ catalyst. In this regard, our group is dealing on how to combine both homogeneous and heterogeneous catalysis to create ‘break through’ catalysts to bridge the gap between both of these aspects. Particularly, we focus on the development of advanced heterogeneous catalytic materials based on single atoms and nanoparticles, which should behave like heterogeneous materials with regard to stability and recycling, and homogeneous complexes with respect to activities and selectivities. In addition to heterogeneous materials, we develop homogeneous catalysts based on organometallic and molecularly defined complexes. Our more prominence is on the preparation of catalysts based on earth abundant metals (Fe, Mn, Co, Ni, Cu) due to their inherited advantages such as more abundance, less expansive and significantly less toxicity. From the past decade, we developed a number of non-noble metal-based single atoms, nanoparticle and molecularly defined catalysts and applied for industrially relevant reactions as well as biomass valorization and upscaling of plastics- and biogenic- waste.
Our selected publications related to these works: Science, 2022, 376, 1433-1441; Science, 2013, 342, 1073-1076; Science, 2017, 358, 326-332; Nature Catalysis 2022, 5, 20- 29; Nature Chemistry, 2013, 5, 537- 543; Chem, 2022, 8, 508-531; Nature Communications, 2025, 16, 3476; Chem, 2026, 12, 102667.
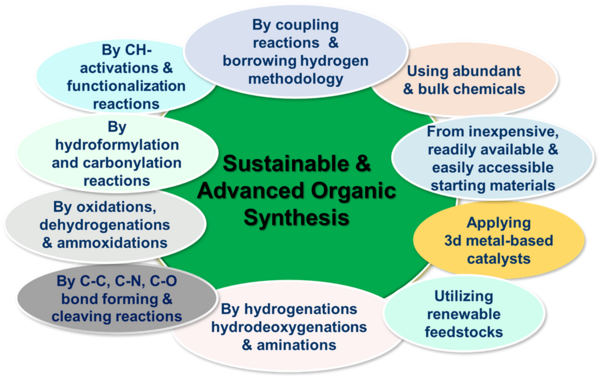
Chemical synthesis (organic synthesis) occupies a unique position in chemistry, the central science, with a profound impact on the modernization of society and human lives. Organic synthesis is an imperative tool that has the power to replicate the fascinating molecules of living nature in the laboratory and industry. These synthetic strategies allow to produce chemical products, materials and life science molecules for potential applications in chemistry, biology, medicine and other areas of science and technology as well as in daily life activities. Considering the importance of organic synthesis, there is a strong demand for the advancement of more sustainable, efficient, selective, cost-effective and innovative organic synthetic methods. This paramount importance stems from the need to move away from traditional methods, which are often inefficient, energy-intensive, and produce hazardous waste. In our group, we are working on the development of more sustainable catalytic methodologies for the synthesis of industrial fine and bulk chemicals, functionalized and structurally diverse molecules, pharmaceuticals, agrochemicals and biomolecules. For these syntheses, we apply catalytic C-N, C-C, C-O and C-X (X=F, Cl and Br) bond forming and cleaving reactions. More specifically, we apply industrially relevant processes such as catalytic hydrogenations, aminations, oxidations, ammoxidations, dehydrogenations, hydrodeoxygenations, coupling reactions as well as CH-activation and functionalization reactions. To carry out these processes, we use inexpensive and readily available starting materials and renewable feedstocks, as well as abundant and more environmentally friendly reagents.
Our selected publications related to these works: Science, 2022, 376, 1433-1441; Science, 2022, 376, 1433-1441; Science, 2013, 342, 1073-1076; Science, 2017, 358, 326-332; Nature Catalysis, 2022, 5, 20-29; Chem, 2022, 8, 508-531; Chem, 2024, 10, 1897-1909.
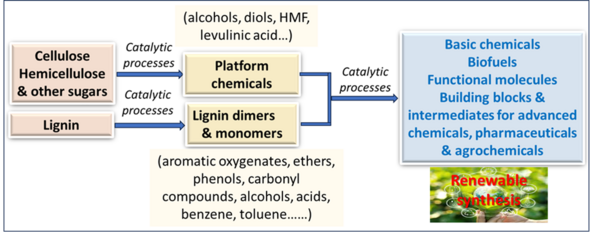
The production of chemicals and energy relies mainly on non-renewable fossil-based feedstocks. However, the future depletion of these fossil-based resources imposed to search for renewable resources as alternatives. The transition to renewable feedstocks offers significant sustainability benefits and supports the development of a circular economy. Thus, effective utilization of renewable resources to produce chemicals, fuels and energy is a major goal of chemical research. Among the different renewable resources, biomass is an organic polymeric material that can be converted into a wide range of higher-value products, including biofuels, biochemicals, and bioplastics. Hence, our work is devoted towards the catalytic valorization of lignin-, cellulose and related sugar-based biomass to value added chemicals, biofuels and functional molecules as well as advanced feedstocks and intermediates for chemical, pharma and agrochemical industrials. Next, we also work on the catalytic conversion of vegetable oils, which are produced on >200 million tons per year from plant sources to essential fatty chemicals such as fatty acids, fatty nitriles, fatty alcohols and fatty amines as well as long chain olefins and alkanes under mild conditions.
Unsere ausgewählten Publikationen zu diesen Werken: Nature Communications, 2025, 16, 3476; JACS, 2025, doi.org/10.1021/jacs.5c10097; Chem Catalysis, 2022, 2, 178-194; Chem, 2024, 10, 1897-1909.

CO2 serves as a renewable, and low-cost resource to produce high-value products. Our interests in CO2 valorization are: (i) hydrogenation of CO2 to syngas (ii) applications of CO2 in carbonylation and hydroformylation processes to produce various industrial chemicals.
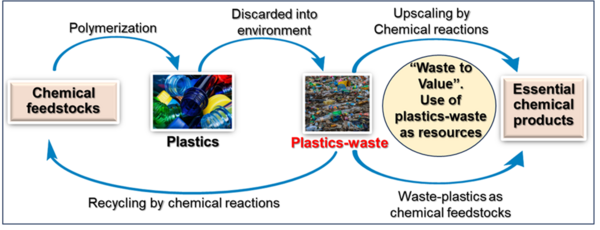
Worldwide, over 15 billion tons of waste are generated annually, causing not only environmental problems but also results in the massive utilization and significant loss of resources. To enable a circular economy, the recycling and reuse of waste materials is crucial for resource conservation, protecting the environment and building a sustainable future. Among the waste materials, about 400 million tons of plastics-waste and 1.3 billion tons of food and organic waste are generated annually, which should be recycled and upscaled to essential products. Our work is particularly directed towards upscaling plastics-waste and use them as resources/feedstocks to produce other value-added products and fuels to enable the concept ‘Waste-to-Value’. Applying suitable catalytic processes, we convert plastics-waste generated from polyethylene, polystyrene, polyester, polyamide and others to produce valuable chemicals as well as fuels. Further, our research program also focuses on the conversion of bio-waste from used cooking oil, food- and agricultural-waste into chemical products, which is of importance in the context of a circular bioeconomy.
Unsere ausgewählten Publikationen zu diesen Werken: Chem, 2026, 12, 102667; JACS, 2026, doi.org/10.1021/jacs.5c10097; Nature Communications, 2025, 16, 3476.
Selected publications
1. V. G. Chandrashekhar, W. Baumann, M. Beller, R. V. Jagadeesh, Science, 2022, 376, 1433-1441. Nickel-catalyzed hydrogenative coupling of nitriles and amines for general amine synthesis.
2. R. V. Jagadeesh, K. Murugesan, A. S. Alshammari, H. Neumann, M.-M. Pohl, J. Radnik, M. Beller, Science, 2017, 358, 326-332. MOF-derived cobalt nanoparticles catalyze a general synthesis of amines.
3. R. V. Jagadeesh, A. E. Surkus, H. Junge, M. M. Pohl, J. Radnik, J. Rabeah, H. Huan, V. Schünemann, A. Brückner, M. Beller, Science, 2013, 342, 1073-1076. Nanoscale Fe2O3-based catalysts for selective hydrogenation of nitroarenes to anilines.
4. Z. Ma, N. Rockstroh, Z. Chen, V. Goyal, C. Kuloor, S. Bartling, Z. Baďura, J. Rabeah, L. Dong, H. Lund, B. Nan, R. Zbořil, R. V. Jagadeesh, M. Beller, Nature Catalysis, 2026 (Accepted). Iron-based single atom catalysts for selective ammoxidation of C(sp3)-H bonds and oxidative C-C cleavage reactions.
5. V. G. Chandrashekhar, T. Senthamarai, R. G. Kadam, O. Malina, J. Kašlík, R. Zbořil, M. B. Gawande, R. V. Jagadeesh, M. Beller, Nature Catalysis, 2022, 5, 20-29. Silica supported Fe/Fe-O nanoparticles for the catalytic hydrogenation of nitriles to amines in the presence of aluminium additives.
6. C. Kuloor, Akash, V. Goyal, R. Zbořil, M. Beller, R. V. Jagadeesh, Angewandte Chemie International Edition, 2025, 64, e202414689. Nickel-catalyzed reductive hydrolysis of nitriles to alcohols.
7. Z. Ma, C. Kuloor, C. Kreyenschulte, S. Bartling, O. Malina, M. Haumann, P. W. Menezes, R. Zbořil, M. Beller, R. V. Jagadeesh, Angewandte Chemie International Edition, 2024, 136, e202407859. Development of iron-based single atom materials for general and efficient synthesis of amines.
8. F. Poovan, R. V. Jagadeesh, M. Beller, Chem, 2026, 12, 102667. A catalytic approach to the valorization of polyesters and biogenic waste for the production of amines.
9. T. Senthamarai, V. G. Chandrashekhar, N. Rockstroh, J. Rabeah, S. Bartling, R. V. Jagadeesh, M. Beller, Chem, 2022, 8, 508-531. A “universal” catalyst for aerobic oxidations to synthesize (hetero)aromatic aldehydes, ketones, esters, acids, nitriles and amides.
10. R. V. Jagadeesh, H. Junge, M. M. Pohl, J. Radnik, A. Brückner, M. Beller, Journal of the American Chemical Society, 2013, 135, 10776-10782. Selective oxidation of alcohols to esters using heterogeneous Co3O4-N@C under mild conditions.
11. B. Zhou, V. Chandrashekhar, Z. Ma, C. Kreyenschulte, S. Bartling, H. Lund, M. Beller, R. V. Jagadeesh, Angewandte Chemie International Edition, 2023, 62, e20221569. Development of a general and selective nanostructured cobalt catalyst for the hydrogenation of benzofurans, indoles and benzothiophenes.
12. K. Murugesan, M. Beller, R. V. Jagadeesh, Angewandte Chemie International Edition, 2019, 58, 5064-5068. Reusable nickel nanoparticles-catalyzed reductive amination for selective synthesis of primary amines.
13. K. Murugesan, Z. Wei, V. G. Chandrashekhar, H. Neumann, A. Spannenberg, H. Jiao, M. Beller, R. V. Jagadeesh, Nature Communications, 2019, 10, 5443.Homogeneous cobalt-catalyzed reductive amination for synthesis of functionalized primary amines.
14. T. Senthamarai, K. Murugesan, J. Schneidewind, N. V. Kalevaru, W. Baumann, H. Neumann, P. C. J. Kamer, M. Beller, R. V. Jagadeesh, Nature Communications, 2018, 9, 4123. Simple ruthenium-catalyzed reductive amination enables the synthesis of a broad range of primary amines.
15. K. Murugesan, V. G. Chandrashekhar, C. Kreyenschulte, M. Beller, R. V. Jagadeesh, Angewandte Chemie International Edition, 2020, 59, 17408-17412. A general catalyst based on cobalt-core-shell nanoparticles for hydrogenation of N-heteroarenes including pyridines.




